A Brief Overview of the History, Use, and Mechanism of Stains
I recently invested in microscopy stains. Some 100 mLs of Lugol’s Iodine and Methylene Blue, nothing too fancy. They got me thinking about microscopy stains as a whole, and, diving into it, they’re fascinating. I want to share some of my findings here.
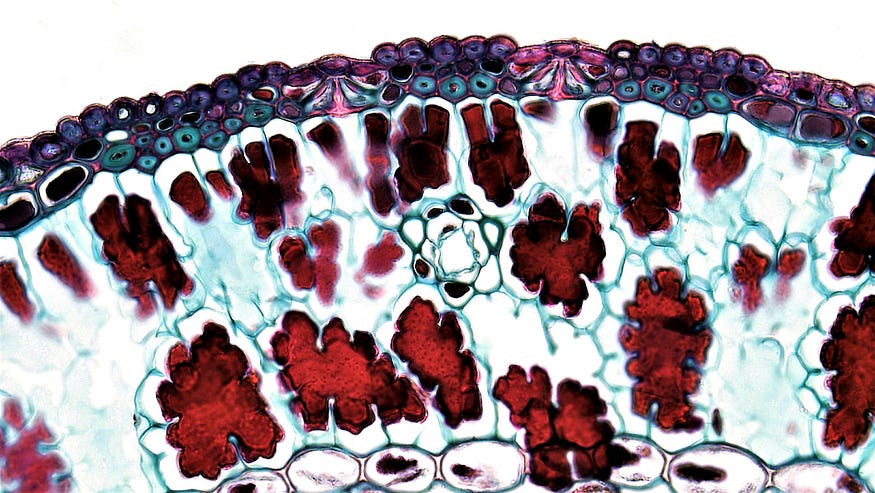
The first use of a microscopy stain is attributed to Anton Van Leeuwenhock, a ‘largely self-taught’ Dutch scientist who, in 1673, used a dye extracted from a saffron crocus bulb and used it to dye his specimens. Van Leeuwenhock is popularly considered the Father of Microbiology, one of the first microscopists and microbiologists, and the first person to view bacteria, protozoa, and a red blood cell under the microscope.
But why did he require a dye in the first place, and one, no less, from the most expensive spice in the world?
Most ‘living’ specimens, such as cells, are typically transparent and colorless. They lack contrast and appear as clear, featureless blobs splattered across a clear, glass surface. Not ideal for observation.
Enter the microscopy stain, a special dye used to ‘color’ the microscope specimens or parts of the specimens. In essence, it is coloring added to the specimen to boost contrast and make structures discernible. But not any random coloring will do. We must use a compound that interacts with the specific specimen in the exact way a microscopist desires.
For Van Leeuwenhock, that one time in 1673, a saffron extract proved best, but modern microscopists have at their disposal a whole arsenal of optimized dyes and stains to beautify their specimens.
A stain is essentially a solution of a salt in a suitable solvent, say alcohol or water. The salt is made up of two halves, a positive cation and a negative anion. One of these ions is colored; it gives the stain its colorful properties and is known as the chromophore. The second uncolored ion is called the counterion.
There are two broad categories of stains: positive stains and negative stains. The majority of dyes used in microscopy are positive stains, and positive stains are almost always basic, which means they have a positive ion as the chromophore. Yes, there are a lot of overlapping categories here, but bear with me, it gets better. Let’s take Methylene Blue as an example.
Methylene blue is a basic stain, meaning it has a positively charged chromophore or coloring ion. These chromophores are attracted to the organelles of cells–membranes, nucleic acids, and cytoplasmic proteins–which are negatively charged. Thus, the stain is absorbed by the cells, and the chromophores stick to the target organelles. This stains the entire cell a signature blue, with bigger organelles that attract more dye, such as a nucleus, stained a deeper blue.
On the flip side are the negative stains, which are typically acidic and have a negative ion as the chromophore; and these work in precisely the opposite manner.
Remember that most organelles in cells are negatively charged. Like charges repel. So the negatively charged chromophores are repelled by the specimen and prevented from entering the specimen. Instead of coloring the cell, these stains color the background (the microscope slide on which the specimen is mounted) while the uncolored specimen stands out as a silhouette.
To summarize, positive stains are basic and have positively charged chromophores, while negative stains are acidic and have negatively charged chromophores. Yes, the terminology tangles up pretty easily. I’ll leave a summary table here to keep things organized.
Both the staining scenarios detailed above are examples of simple staining, where one stain is used and all organisms are stained the same color. Methylene blue is a non-specific stain. It will bind to negatively charged organelles nondiscriminately. It is best used when you have only one type of specimen on the slide or you only need to observe the morphology, size, or arrangement of the cells.
One level of complexity above is differential staining, which is able to differentiate between different types of organisms. This technique utilizes multiple stains and different specimens assume different colors based on their specific interactions with the chromophores.
One well-known example of differential staining is Gram staining, which is used to differentiate bacteria based on the variations in their cell walls. Gram staining uses crystal violet as a primary stain and safranin as a counterstain. A counterstain is a secondary stain used to enhance contrast, in this case between the different types of bacteria. It also requires alcohol as a decolorizer (a substance that removes dyes) and Lugol’s Iodine as a mordant. A mordant is a chemical agent with the power of making dyes stain otherwise unstainable materials. I won’t go through the entire process of gram-staining here, but here’s a link for those interested.
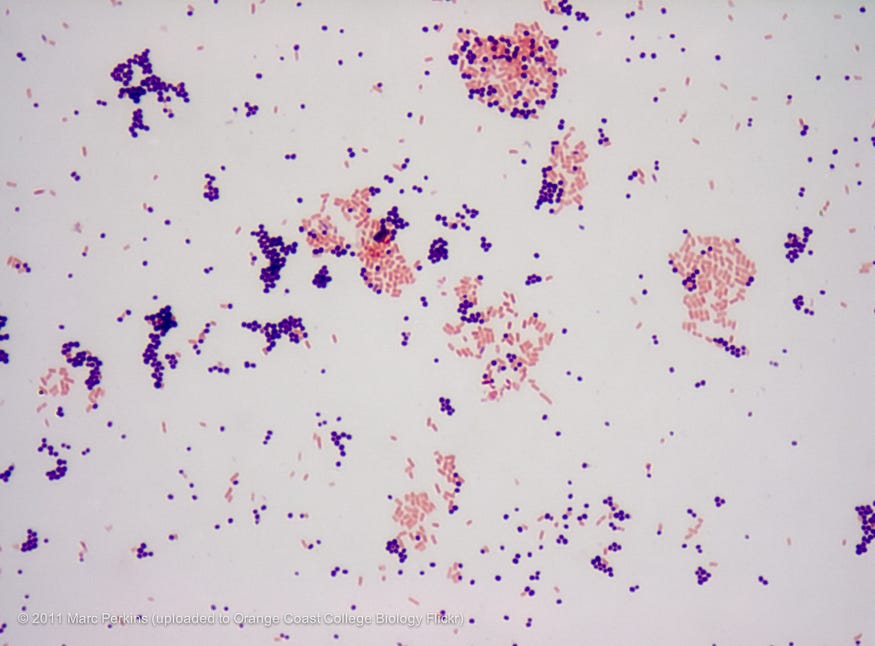
Stains can be further used to differentiate between live and dead cells, as some stains are only taken up by living cells while others are only by dead cells. They can be used to highlight metabolic activity, classify microorganisms, for medical diagnosis, and even for medicinal uses. Microscopy has drastically improved since the 1670s, and stains are often now used in complex and brilliant ways for niche applications. There is also, of course, fluorescence microscopy, phase contrast, DIC, darkfield, electron microscopy, and more for more advanced analysis.
I’ve only just scratched the surface here. The rabbit hole extends far deeper. Maybe I’ll let myself fall further in someday, but as for now, I’m twitching to get my (gloved) hands dirty with my new stains. Keep a lookout for that.
This is not a tutorial for using microscopy stains. All stains listed in this article are toxic and must be handled properly. Read the MSDS before handling any laboratory chemicals, and don’t be stupid.
Sources: See here
